Ethylene sulfate is an important organic chemical compound that belongs to the class of cyclic sulfates. It is mainly used in industrial and research applications, especially in organic synthesis and electrochemistry. This compound has gained attention due to its role as an intermediate in various chemical processes and its relevance in advanced material development. Understanding ethylene sulfate is essential for chemists and industries that deal with electrolyte formulations, polymer chemistry, and specialty chemicals.
What is Ethylene Sulfate
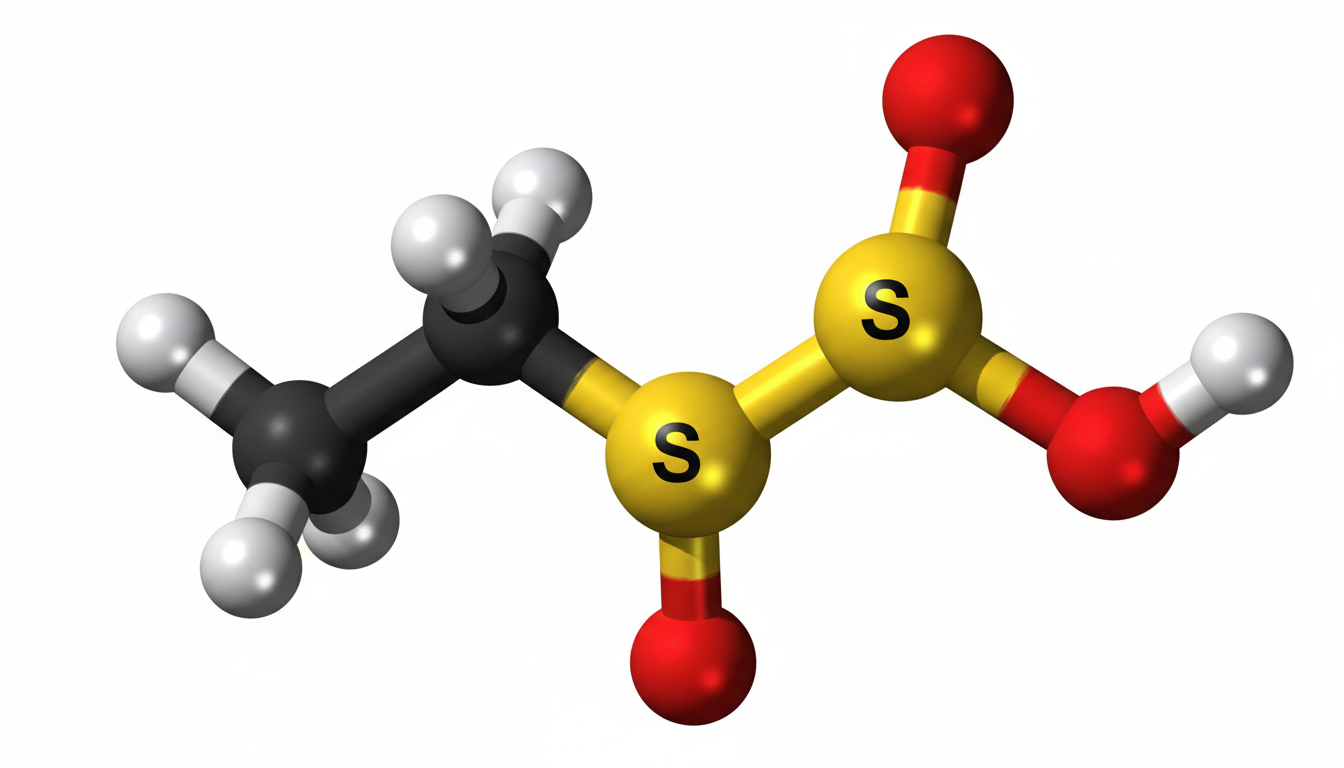
Ethylene sulfate is a cyclic ester derived from ethylene glycol and sulfuric acid. It contains a strained ring structure that makes it highly reactive under certain chemical conditions. Because of this reactivity, it is commonly used as an intermediate rather than a final product in chemical manufacturing. The compound is generally studied in controlled laboratory environments due to its sensitivity and potential to undergo hydrolysis and other reactions.
The molecular structure of ethylene sulfate allows it to interact easily with nucleophiles, making it valuable in synthesis pathways. It is often used in reactions where controlled ring-opening is required to form more complex organic molecules.
Chemical Structure and Properties
Ethylene sulfate has a cyclic structure containing carbon, hydrogen, oxygen, and sulfur atoms. The sulfur atom is double-bonded to oxygen atoms, forming a sulfate group within a ring system. This ring strain contributes to its chemical reactivity.
Physically, ethylene sulfate is typically a colorless or pale liquid or solid depending on conditions. It is not highly stable in the presence of moisture and tends to decompose in water, producing ethylene glycol derivatives and sulfur-containing byproducts.
Chemically, it behaves as an electrophilic compound. It reacts readily with nucleophiles such as water, alcohols, and amines. This makes it useful in controlled chemical synthesis processes. Its reactivity is one of the main reasons it must be handled with care in laboratory settings.
Synthesis of Ethylene Sulfate
Ethylene sulfate is commonly synthesized through the reaction of ethylene oxide or ethylene glycol with sulfur trioxide or chlorosulfonic acid under controlled conditions. The reaction involves the formation of a cyclic sulfate ester through intramolecular cyclization.
The synthesis process requires strict temperature control because excessive heat can lead to unwanted side reactions or decomposition. In industrial settings, specialized equipment is used to ensure safe handling of reactive intermediates.
Purification of ethylene sulfate is also important because impurities can affect its performance in downstream applications. Distillation or crystallization methods are often employed depending on the production scale.
Applications of Ethylene Sulfate
Ethylene sulfate is widely used as an intermediate in organic synthesis. It plays a role in the production of specialty chemicals, pharmaceuticals, and advanced materials. One of its most important applications is in electrochemistry, where it is used in electrolyte formulations for batteries.
In battery technology, ethylene sulfate is studied for its ability to improve the stability and performance of electrolytes. It contributes to the formation of stable solid-electrolyte interfaces, which are crucial for efficient energy storage systems.
It is also used in polymer chemistry, where it acts as a modifying agent in the production of certain polymers. Its reactive sulfate ring allows it to participate in chain modification reactions, improving material properties such as strength and flexibility.
In research laboratories, ethylene sulfate is used as a reagent for studying ring-opening reactions and reaction mechanisms involving cyclic sulfate esters.
Industrial Importance
The industrial importance of ethylene sulfate lies in its versatility as a chemical intermediate. It is not typically used in final consumer products but plays a critical role in the synthesis of high-value compounds.
Industries that focus on advanced materials, energy storage, and specialty chemicals rely on compounds like ethylene sulfate for innovation. Its role in improving battery electrolytes has made it particularly relevant in the growing electric vehicle and renewable energy sectors.
Because of its reactivity, it allows chemists to design more efficient and selective chemical pathways, reduce waste and improve production efficiency.
Safety and Hazards
Ethylene sulfate must be handled with care due to its reactive nature. It can cause irritation upon contact with skin, eyes, or respiratory tissues. Inhalation or direct exposure should be avoided, and proper protective equipment is necessary when working with it.
Since it reacts with water, accidental exposure to moisture can lead to decomposition and release of potentially harmful byproducts. Therefore, it is typically stored in dry, sealed containers under controlled conditions.
Laboratory safety procedures such as the use of fume hoods, gloves, and goggles are essential when handling ethylene sulfate. Proper training is also required for personnel working with this compound.
Environmental Impact
The environmental impact of ethylene sulfate depends on how it is managed and disposed of. Because it is chemically reactive, it should not be released into natural water systems or soil. It can undergo hydrolysis, but uncontrolled release may still lead to harmful chemicals by products.
Industries using ethylene sulfate are required to follow strict environmental regulations to prevent contamination. Waste management includes neutralization and controlled decomposition practices before disposal.
Research is ongoing to find safer alternatives and greener synthesis methods to reduce the environmental footprint of sulfate compounds.
Surgery
Ethylene sulfate is a highly reactive and important chemical intermediate used in various industrial and research applications. Its role in synthesis, polymer chemistry, and especially battery technology makes it a valuable compound in modern science. However, due to its reactivity and potential hazards, it must be handled with strict safety measures. As technology advances, ethylene sulfate will continue to play a role in the development of new materials and energy solutions while also encouraging safer and more sustainable chemical practices.