The global pyrogen testing market is witnessing steady growth due to the increasing demand for safe pharmaceutical and medical products. Pyrogen testing plays a crucial role in detecting bacterial endotoxins and ensuring that drugs, vaccines, and medical devices are free from contaminants that can cause adverse reactions in patients. With stringent regulatory requirements and rising focus on product safety, pharmaceutical and biotechnology companies are increasingly adopting advanced pyrogen testing methods.
The growing prevalence of chronic diseases, expansion of the biopharmaceutical sector, and rising investments in healthcare infrastructure are further driving the market. Additionally, the shift toward alternative testing methods such as in vitro assays is gaining traction, replacing traditional animal-based testing methods.
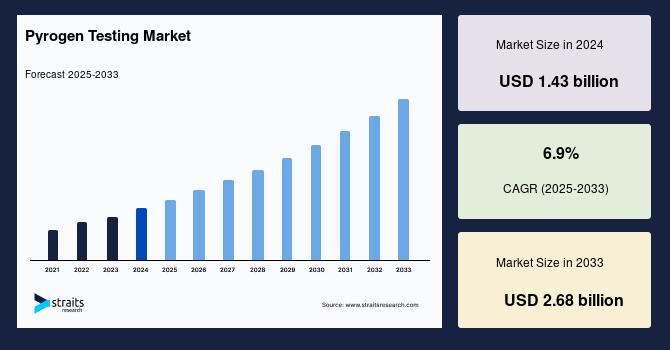
Market Size 2024 – USD 1.43 Billion.
Market Size 2025 – USD 1.57 Billion.
Market Size 2033 – USD 2.68 Billion.
CAGR (2025–2033) – 6.9%.
Get Your Sample Report Here: https://straitsresearch.com/report/pyrogen-testing-market/request-sample
Get Full Report Now: https://straitsresearch.com/report/pyrogen-testing-market
Market Drivers
Increasing Demand for Pharmaceutical Safety
The rising need for safe and effective pharmaceutical products is a major driver of the pyrogen testing market. Regulatory authorities require strict testing of drugs and medical devices to ensure they are free from pyrogens, which can cause fever and other severe reactions in patients.
Growth of Biopharmaceutical Industry
The expansion of the biopharmaceutical sector is significantly contributing to market growth. With the increasing production of biologics, vaccines, and injectable drugs, the demand for pyrogen testing is rising to maintain product safety and compliance.
Shift Toward In Vitro Testing Methods
There is a growing trend toward adopting in vitro testing methods such as the Limulus Amebocyte Lysate test. These methods offer faster, more accurate, and ethical alternatives to traditional animal-based testing, driving their adoption across the industry.
Stringent Regulatory Frameworks
Regulatory bodies worldwide have established strict guidelines for pyrogen testing, ensuring the safety of pharmaceutical and medical products. Compliance with these regulations is encouraging companies to invest in advanced testing technologies.
Market Challenges
High Cost of Advanced Testing Methods
The implementation of advanced pyrogen testing technologies can be expensive, especially for small and medium-sized enterprises. High costs associated with equipment and skilled personnel may limit adoption.
Regulatory Complexity
Navigating regulatory requirements across different regions can be challenging for companies. Variations in standards and approval processes can delay product launches and increase operational costs.
Limited Awareness in Emerging Markets
In some developing regions, limited awareness about advanced pyrogen testing methods and lack of infrastructure may hinder market growth.
Market Segmentation
By Product Type
Instruments
Instruments used in pyrogen testing include specialized equipment designed to detect endotoxins accurately. These instruments are widely adopted in pharmaceutical and biotechnology companies.
Consumables
Consumables such as reagents and test kits hold a significant share of the market due to their recurring usage in testing procedures.
Services
Testing services are gaining popularity as companies outsource their testing requirements to specialized laboratories to reduce costs and improve efficiency.
By Test Type
Limulus Amebocyte Lysate Test
This test is widely used for detecting endotoxins and is considered a standard method in the industry due to its high sensitivity and reliability.
Rabbit Pyrogen Test
The rabbit pyrogen test is a traditional method used to detect pyrogens. However, its usage is declining due to ethical concerns and the availability of alternative methods.
Monocyte Activation Test
This test is gaining traction as an alternative to animal-based testing, offering improved accuracy and compliance with regulatory standards.
By End-User
Pharmaceutical and Biotechnology Companies
These companies are the largest end-users of pyrogen testing solutions, ensuring product safety and regulatory compliance.
Medical Device Companies
Medical device manufacturers rely on pyrogen testing to ensure their products are safe for use and free from contaminants.
Contract Research Organizations
CROs provide testing services to pharmaceutical and biotechnology companies, supporting drug development and regulatory compliance.
Top Players Analysis
- Charles River Laboratories International, Inc.
The company offers a wide range of endotoxin and pyrogen testing solutions, leveraging its strong expertise in laboratory services and regulatory compliance. - Lonza Group AG
Lonza is a key player in the market, providing advanced testing products and services with a focus on innovation and quality. - Merck KGaA
Merck offers comprehensive pyrogen testing solutions, including reagents and instruments, supported by its strong global presence. - Thermo Fisher Scientific Inc.
The company provides a broad portfolio of testing products and services, catering to the needs of pharmaceutical and biotechnology industries. - BioMérieux SA
BioMérieux focuses on diagnostic solutions and offers advanced testing technologies to ensure product safety. - Wako Chemicals USA, Inc.
The company specializes in endotoxin detection products and is known for its high-quality testing solutions. - Associates of Cape Cod, Inc.
This company is recognized for its expertise in endotoxin testing and provides a wide range of products and services. - GenScript Biotech Corporation
GenScript offers innovative testing solutions and supports research and development activities in the pharmaceutical sector.
Future Outlook
The pyrogen testing market is expected to experience significant growth in the coming years, driven by increasing demand for safe pharmaceutical products and advancements in testing technologies. The adoption of in vitro methods is likely to continue, reducing reliance on animal-based testing and improving efficiency.
Additionally, the expansion of healthcare infrastructure in emerging markets and rising investments in research and development will create new opportunities for market players. Strategic collaborations and technological innovations will further enhance market growth.
FAQs
What is pyrogen testing?
Pyrogen testing is a process used to detect fever-causing substances in pharmaceutical products and medical devices.
Why is pyrogen testing important?
It ensures the safety of drugs and medical devices by identifying harmful contaminants that can cause adverse reactions.
Which test method is widely used?
The Limulus Amebocyte Lysate test is widely used due to its accuracy and reliability.
What are the key growth drivers?
Key drivers include rising pharmaceutical safety requirements, growth of the biopharmaceutical industry, and technological advancements.
What challenges does the market face?
Challenges include high costs, regulatory complexities, and limited awareness in certain regions.
About Us
Straits Research is a market intelligence company providing global business information reports and services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insight for thousands of decision-makers. Straits Research Pvt. Ltd. provides actionable market research data, especially designed and presented for decision making and ROI.