The global drug repurposing market is gaining significant traction as pharmaceutical companies seek cost-effective and time-efficient alternatives to traditional drug development. Drug repurposing, also known as drug repositioning, involves identifying new therapeutic uses for existing drugs. This strategy reduces development timelines, lowers risks, and improves the success rate of clinical trials. Increasing demand for faster drug approvals, rising prevalence of chronic diseases, and growing investments in pharmaceutical R&D are driving the market forward. Additionally, advancements in artificial intelligence and bioinformatics are enabling researchers to identify new applications for existing drugs more efficiently.
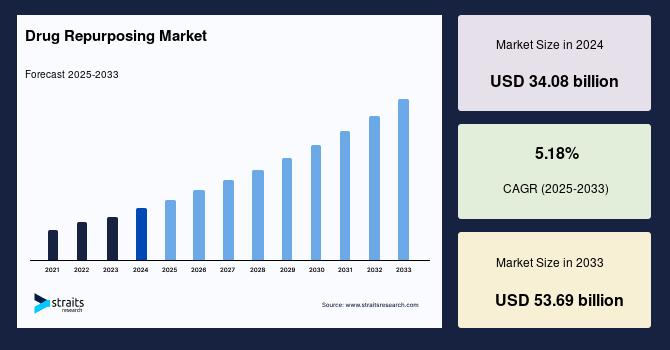
Market Size 2024 – USD 34.08 Billion.
Market Size 2025 – USD 35.84 Billion.
Market Size 2033 – USD 53.69 Billion.
CAGR (2025–2033) – 5.18%.
Get Your Sample Report Here: https://straitsresearch.com/report/drug-repurposing-market/request-sample
Get Full Report Now: https://straitsresearch.com/report/drug-repurposing-market
Market Drivers
Rising Prevalence of Chronic Diseases
The growing burden of chronic diseases such as cancer, cardiovascular disorders, and neurological conditions is a major driver for the drug repurposing market. Existing drugs with established safety profiles are increasingly being evaluated for new therapeutic indications, accelerating treatment availability.
Cost and Time Efficiency in Drug Development
Traditional drug development is expensive and time-consuming, often taking over a decade and billions of dollars. Drug repurposing significantly reduces both cost and time, as safety and pharmacokinetics data are already available. This advantage is encouraging pharmaceutical companies to adopt repurposing strategies.
Technological Advancements in AI and Data Analytics
The integration of artificial intelligence and machine learning in drug discovery is transforming the repurposing landscape. Advanced computational tools help identify potential drug candidates, analyze biological pathways, and predict efficacy, improving success rates.
Increasing Regulatory Support
Regulatory authorities are supporting drug repurposing initiatives by providing incentives such as orphan drug designations and accelerated approval pathways. This has encouraged companies to explore repurposing opportunities.
Market Challenges
Limited Patent Protection
One of the major challenges in drug repurposing is limited intellectual property protection. Since the original drug may already be off-patent, companies may face difficulties in securing exclusivity, reducing financial incentives.
Scientific and Clinical Validation Issues
Although drug repurposing reduces development time, it still requires rigorous clinical trials to validate efficacy for new indications. Unexpected outcomes and lack of efficacy in new therapeutic areas can hinder progress.
Regulatory Complexities
Navigating regulatory frameworks for repurposed drugs can be complex, especially when the new indication differs significantly from the original use. This can delay approvals and increase costs.
Market Segmentation
By Drug Molecule Type
Small Molecules
Small molecules dominate the market due to their well-established pharmacological profiles and ease of modification. These drugs are widely used in repurposing studies because of their known safety and efficacy.
Biologics
Biologics are gaining attention in drug repurposing due to their targeted mechanisms of action. Advances in biotechnology are expanding their applications in treating complex diseases.
By Therapeutic Area
Oncology
Oncology holds a significant share in the drug repurposing market. Many existing drugs are being evaluated for cancer treatment, driven by the urgent need for effective therapies.
Neurological Disorders
Neurological conditions such as Alzheimer’s and Parkinson’s disease are key areas for drug repurposing. Existing drugs are being tested for their potential to address unmet medical needs.
Infectious Diseases
Drug repurposing has played a crucial role in addressing infectious diseases by rapidly identifying potential treatments during outbreaks.
Cardiovascular Diseases
Repurposed drugs are being explored for cardiovascular conditions to improve patient outcomes and reduce mortality rates.
By End-User
Pharmaceutical and Biotechnology Companies
These companies are the primary contributors to the market, investing heavily in research and development to identify new drug applications.
Research Institutes
Academic and research institutions play a critical role in discovering new therapeutic uses for existing drugs through collaborative studies.
Contract Research Organizations
CROs support drug repurposing initiatives by providing clinical trial and research services, enabling faster development processes.
Top Players Analysis
- Pfizer Inc.
Pfizer is actively involved in drug repurposing initiatives, leveraging its extensive drug portfolio and advanced research capabilities to identify new therapeutic applications. - Novartis AG
Novartis focuses on innovative research and strategic collaborations to expand the use of existing drugs across multiple therapeutic areas. - Johnson & Johnson
The company invests in advanced technologies and partnerships to accelerate drug repurposing and improve patient outcomes. - Merck & Co., Inc.
Merck emphasizes data-driven approaches and clinical research to explore new indications for established drugs. - GlaxoSmithKline plc
GSK is involved in repurposing efforts, particularly in infectious diseases and oncology, utilizing its strong R&D infrastructure. - Teva Pharmaceutical Industries Ltd.
Teva focuses on generic drugs and repurposing strategies to maximize the value of its existing portfolio. - F. Hoffmann-La Roche Ltd
Roche leverages precision medicine and advanced diagnostics to identify new uses for its drug portfolio. - Sanofi S.A.
Sanofi invests in innovative research to expand the applications of its existing drugs, particularly in rare diseases.
Future Outlook
The drug repurposing market is expected to witness steady growth over the forecast period. Increasing collaborations between pharmaceutical companies, research institutes, and technology providers will drive innovation. The adoption of AI and big data analytics will further enhance the efficiency of identifying repurposing opportunities. Moreover, the rising focus on personalized medicine and targeted therapies will create new growth avenues. Emerging markets are also expected to contribute significantly due to increasing healthcare investments and demand for cost-effective treatments.
FAQs
What is drug repurposing?
Drug repurposing is the process of identifying new therapeutic uses for existing drugs that are already approved or under development.
Why is drug repurposing important?
It reduces drug development time, lowers costs, and improves the success rate of clinical trials.
Which therapeutic area dominates the market?
Oncology is one of the leading segments due to the high demand for effective cancer treatments.
What are the key growth drivers?
Key drivers include rising chronic diseases, cost efficiency, technological advancements, and regulatory support.
What challenges does the market face?
Challenges include limited patent protection, regulatory complexities, and the need for clinical validation.
About Us
Straits Research is a market intelligence company providing global business information reports and services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insight for thousands of decision-makers. Straits Research Pvt. Ltd. provides actionable market research data, especially designed and presented for decision making and ROI.